Stirrups & Stirrup Solutions
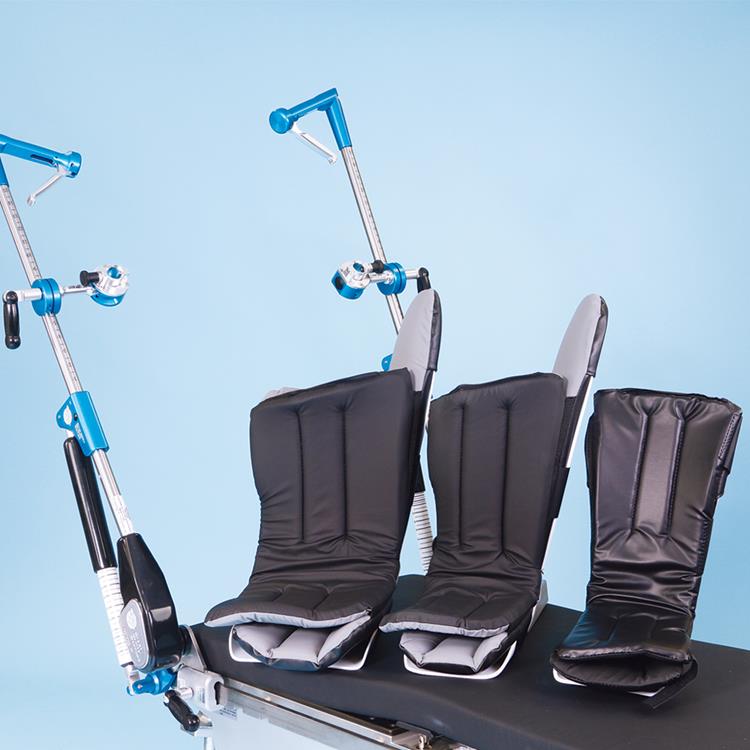
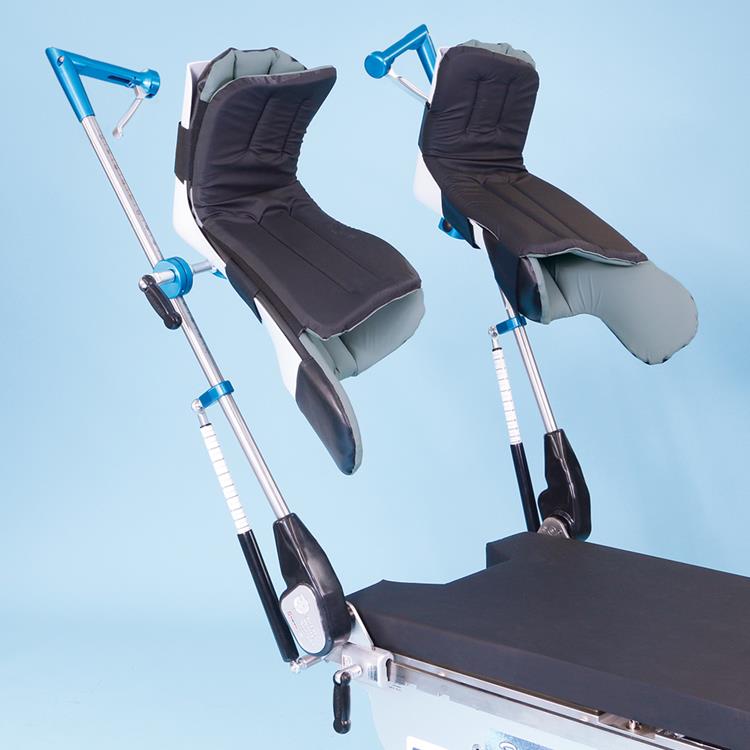
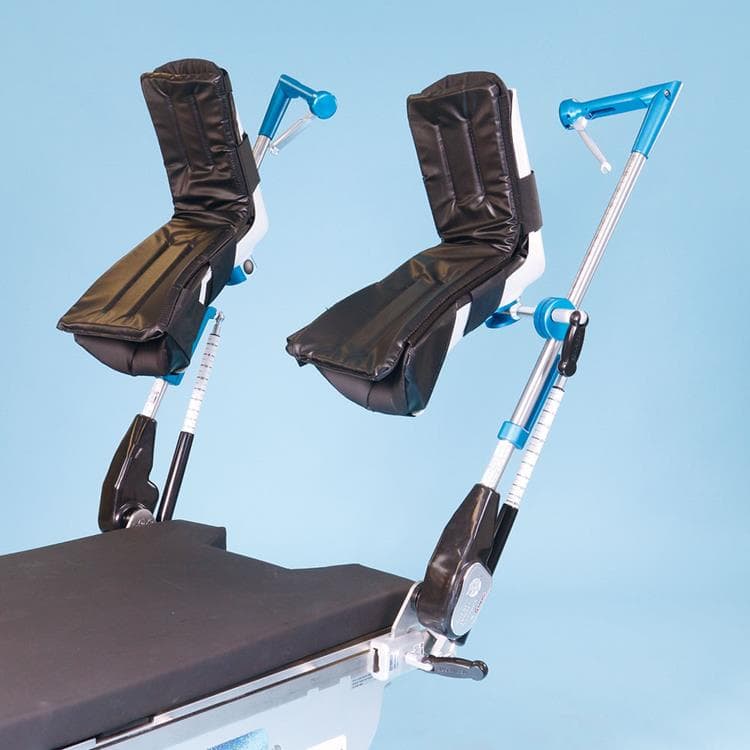
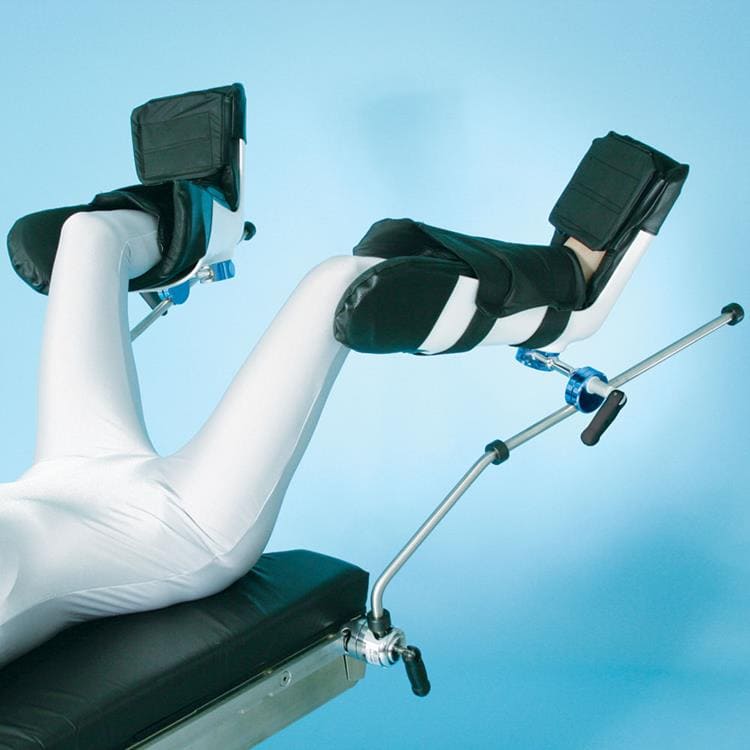
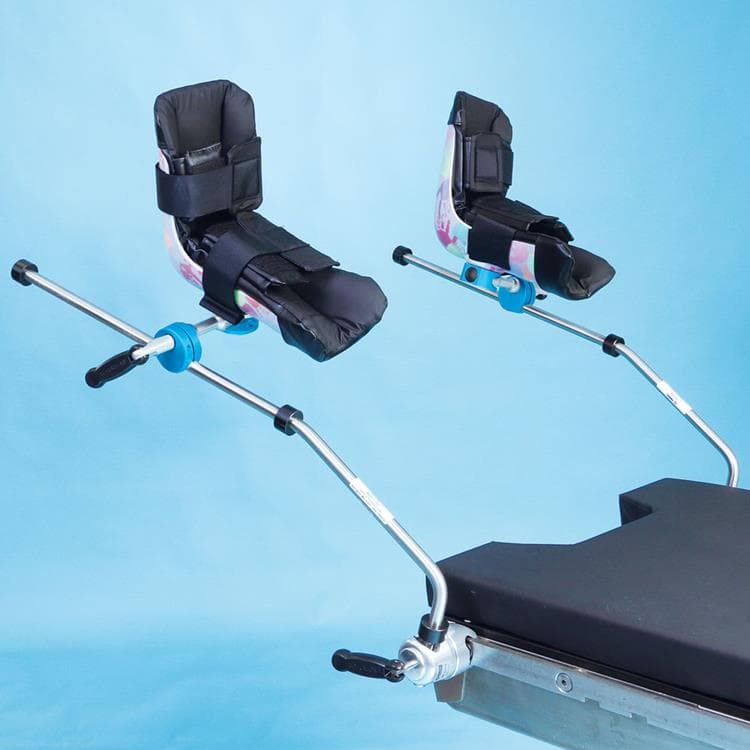
Great White Platinum Stirrups
The Great White Platinum Stirrups deliver precision surgical support with advanced, three-axis control that mirrors the natural movement of the human hip. The surgeon simply squeezes the Trigger-Grip handle to smoothly adjust lithotomy (−30° to +75°), adduction/abduction (−9° to +25°), and external rotation (20°) while maintaining sterility and patient comfort. Upon release, the stirrup locks securely in any position. Built-in lithotomy and length indicators aid rapid repositioning, and extended lateral boot fins help minimize superficial nerve pressure. With a robust 600-lb patient weight capacity, these fully assembled stirrups include deluxe boot liners and mount swiftly via side-rail blade clamps.
Great White HD1000 Stirrups
The Great White HD1000 introduces the world’s first interchangeable boot system, enabling seamless transitions among Premium, Platinum, and Bariatric configurations. It upholds three-axis control and a generous range of motion (−35° to +85° lithotomy; −9° to +25° adduction/abduction). Supplied with three boot types—Premium, Platinum, and Bariatric—the HD1000 supports an impressive 1000-lb capacity, delivering cost-effective versatility across procedures.
Great White Maxima Stirrups
Designed for bariatric applications, the Great White Maxima Stirrups offer the same three-axis control and smooth Trigger-Grip maneuverability across a −30° to +80° lithotomy span and −9° to +25° adduction/abduction range. Built to accommodate up to 800 pounds, these heavy-duty platforms feature an extended lateral boot design to reduce peroneal nerve risk and include deluxe boot liners for secure patient support.
Great White Robotic Stirrups
Optimized for robotic gynecologic and urologic surgery, the Great White Robotic Stirrups provide ultra-precise, intra-operative leg control. Mirroring the Platinum line’s three-axis mechanics, they offer motion from −65° low to +45° high lithotomy, with −9° to +25° adduction/abduction and 20° external rotation. These stirrups include premium boot pads, support up to 600 pounds, and mount via side-rail blade clamps for reliable integration into robotic workflow.
Great White Premium Stirrups
The Great White Premium Stirrups provide streamlined, reliable intra-operative positioning with three-axis control and smooth Trigger-Grip handling. Covering a lithotomy range of −30° to +80° and adduction/abduction of −9° to +25°, these stirrups include durable boot pads and leg indicators for precision. They are a high-quality solution at a value-optimized price point.
Pediatric E-Z Lift Stirrups
Tailored for pediatric patients, the Pediatric E-Z Lift Stirrups offer intuitive control with axial rotation via a single clamp boot that simplifies positioning. The floating boot adapts dynamically while repositioning. These stirrups handle abduction (+25°), adduction (−9°), lithotomy from +84° to −35°, and 20° external rotation. Available in PUPS (160-lb) and KIDS (250-lb) models, they provide safe, responsive support for young patients.
Lithotomy Stirrups
The Lithotomy Stirrups offer secure leg support throughout gynecologic, laparoscopic, or urologic procedures. Featuring an extended lateral boot design and soft boot pads, they prevent leg rotation and reduce superficial nerve risk. With a 350-lb working capacity and swift side-rail attachment, they provide dependable stability for prolonged operations.
Pediatric Lithotomy Stirrups
Specifically designed for pediatric use, the Pediatric Lithotomy Stirrups securely encapsulate the lower leg with easy-clean liners. The floating boot maintains adaptability during repositioning. Available in PUPS (160 lbs) and KIDS (250 lbs), with float adjustment across abduction (+25°) and adduction (−9°), plus lithotomy movements from +84° to −35°, they ensure precision positioning for young surgical patients.
Candy Cane Stirrups
The Candy Cane Stirrups represent a cost-effective, traditional positioning solution. Constructed from sturdy stainless steel, they offer height adjustability (28″–44″) with 8″ leg clearance and include durable double-loop ankle straps. Their ergonomic twist-lock handle ensures precise control—ideal for general surgical applications where efficiency and simplicity are paramount.
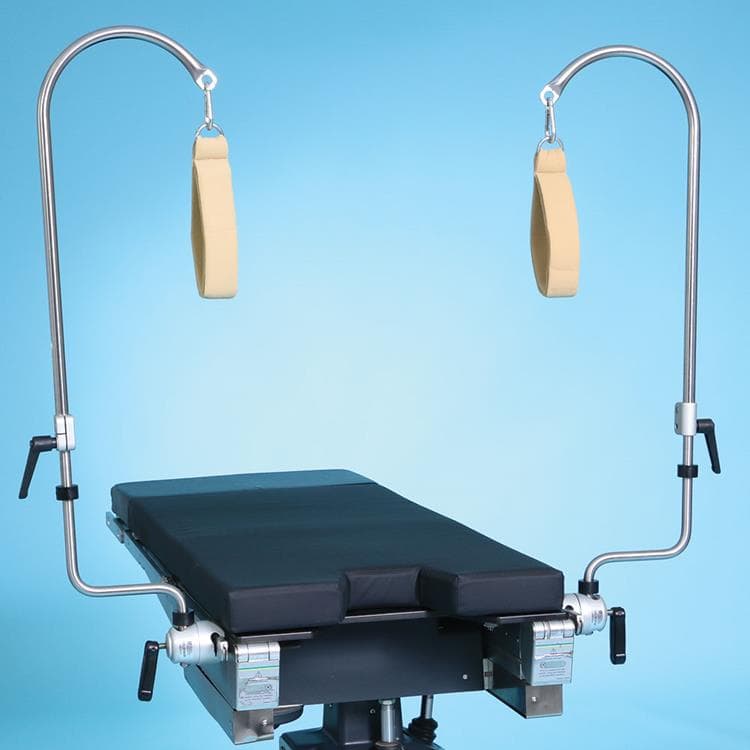
Nissen Straps
Nissen Straps are engineered to secure patient legs during steep reverse Trendelenburg positioning. Each leg strap includes a 1″ thick pad, spans 48″, and supports up to 600 pounds, with quick side-rail mounting. Bariatric versions stretch to 72″ and support 800 pounds, offering expanded safety for larger patients.
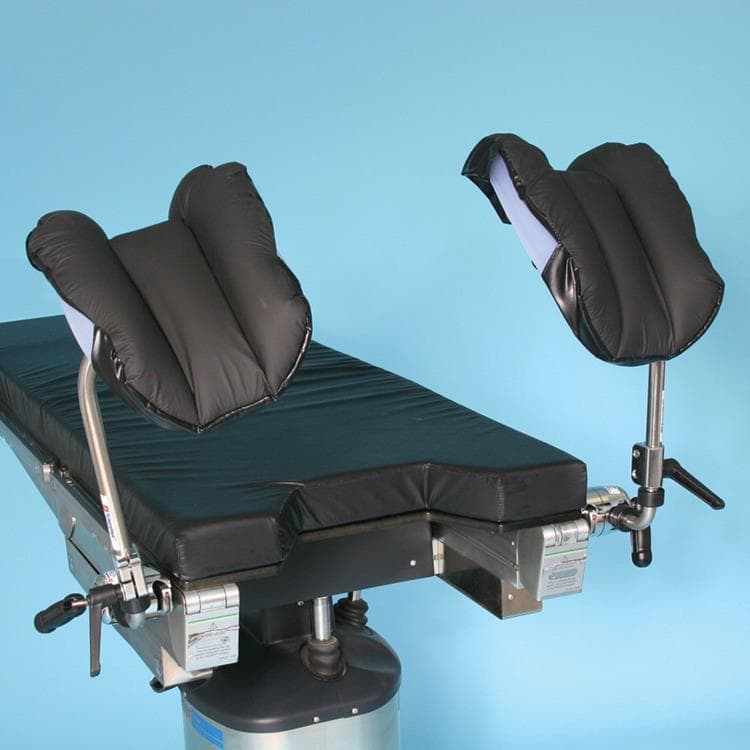
Knee Crutches
The Knee Crutches afford adjustable stability with a vertical range of 12″ to 19″ and 360° rotation via an ergonomic locking handle. Built from premium stainless steel and furnished with soft liners to minimize nerve pressure, they mount easily using Schure Socket XL clamps for versatile intra-operative support.
Uro-Trapper System
The Uro-Trapper System delivers advanced fluid containment without impeding patient access. This flexible drape support attaches securely to any surgical table and maintains optimal fluid drainage—even after compression—by springing back into position. Designed for hysteroscopy, cystoscopy, urologic, and oncologic procedures, its intuitive clamps simplify setup amid high-tech operative environments.