SchureSpine Frame

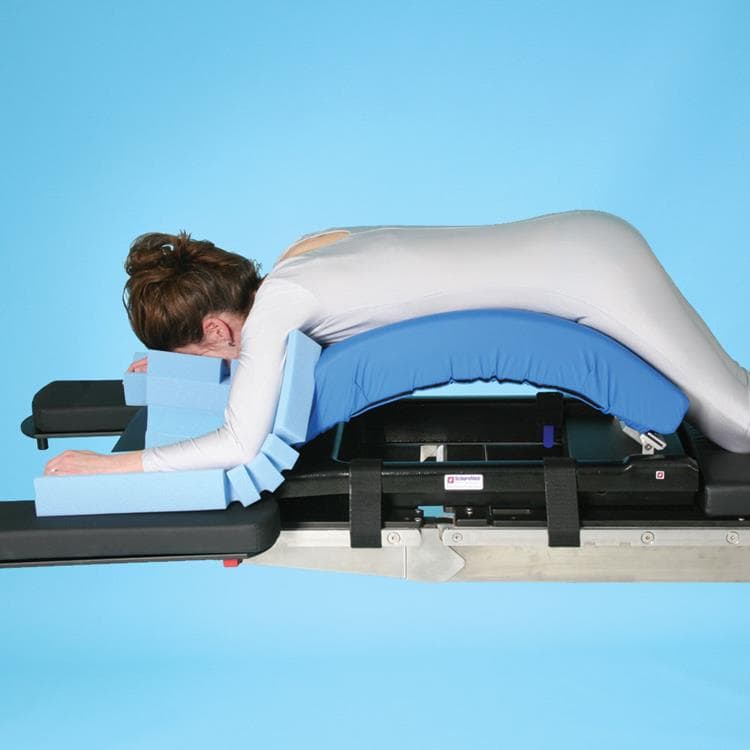
SchureMed SchureSpine Frame
(800-0235)
The Schure Spine Frame provides a stable and convenient platform for spine surgeries such as laminectomies, decompressions, and disc surgery. You can adjust it intraoperatively to the desired lordosis to get optimal surgical site exposure. Tapered adjustable pads provide optimal patient safety and comfort.
The Schure Spine Frame has improved features over the current Wilson™ Frame, has a patient weight capacity of 500 lbs. (227 kg) and is C-arm compatible. It can be used with any O.R. table, and fits the Jackson Spine Table and Allen Flex Frame as well. Comes complete with transport storage dolly, disposable headrest and 2 arm cradle pads (retail customers only).
- Adjusts intra-operatively to the desired lordosis to get optimal surgical site exposure
- Transport dolly of durable stainless steel included
- Patient weight capacity is 500 lbs. (227 kg)
- Schure Spine Frame Pad Set and Crank Handle included
Optional Accessories
#508-0373 Disposable Laminectomy Arm Cradles
#508-1340 Disposable Richards Slotted Headrests, Extended
#800-0074-SF Schure Spine Frame Dolly
Replacement Products
#508-0472 Schure Spine Frame Pad Set
#800-0277 Schure Spine Frame Crank Handle

